
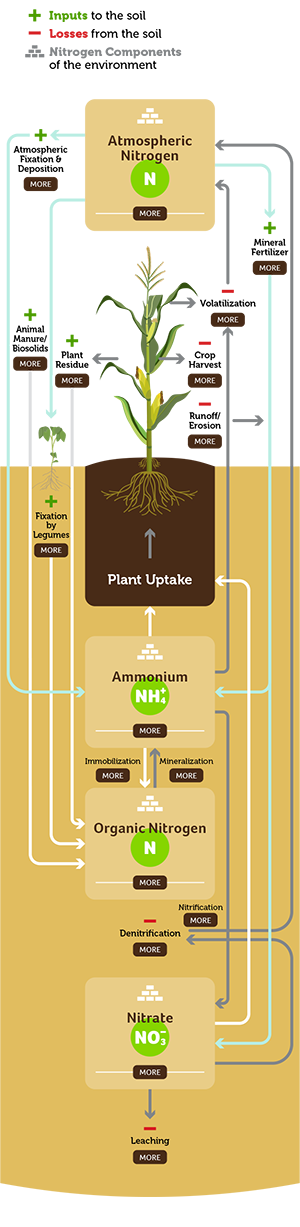
Atmospheric Nitrogen
Nitrogen gas in the atmosphere represents the largest quantity of the nutrient in the environment but is unavailable to most plants as a direct source. Nitrogen gas is used to manufacture nitrogen fertilizers or can be “fixed” by certain soil organisms.
Atmospheric Fixation & Deposition
Atmospheric nitrogen gas (N2) must be processed or “fixed” into nitrates or nitrites to be usable by plants. When more nitrogen is fixed from the atmosphere than the plant needs, excess atmospheric nitrogen is deposited in the ecosystem.
Mineral Fertilizers +
Most of the nitrogen in commercially available fertilizer is derived by combining atmospheric N2 with H2 to form ammonia (NH3), which can be used as fertilizer (anhydrous ammonia) or further processed into other dry or liquid nitrogen fertilizers such as urea, ammonium sulfate, or liquid nitrogen fertilizers.
Volatilization -
This happens when ammonia gas (NH3) is lost to the atmosphere from anhydrous ammonia, urea, or liquid nitrogen fertilizer sources. These losses can be minimized by proper fertilizer placement below the soil surface.
Animal Manure/Biosolids +
Animal manure is an important source of nitrogen – the quantity of nitrogen in the manure depends on the animal species, their age and diet, and bedding material.
Plant Residues +
Small quantities of soil nitrogen are contained in plant residues remaining after harvest. Nitrogen in plant residues becomes available as the residues are decomposed by soil organisms.
Crop Harvest -
This represents the amount of nitrogen that’s in the harvested portion of the crop and removed from the field completely. Use our nutrient removal calculator to find out much nitrogen, and other nutrients, you should be replacing.
Runoff/Erosion -
Water that doesn’t infiltrate soil and is lost by surface flow is runoff. Erosion refers to the wearing away of soil through rain, irrigation, wind, and ice. Both these actions result in lost nutrients, including nitrogen, from the soil. Runoff and erosion can be minimized by maintaining soil cover with crop residue and/or cover crops.
Fixation by Legumes +
Legumes such as soybeans and alfalfa “fix” nitrogen by converting atmospheric N2 to plant-available forms in a process that involves rhizobium bacteria and plant roots. This fixed nitrogen can provide nitrogen for the next crop or be removed in harvested plant material. Legumes usually do not need supplemental fertilizer nitrogen.
Denitrification -
When finer-textured soil becomes saturated, some organisms look for oxygen by decomposing NO3 – a process called denitrification. The NO3 is converted to gases that are unavailable to plants and can cause significant losses of nitrogen when soil is warm and remains saturated. Denitrification losses can be minimized by split applications to avoid high soil nitrogen during wet weather.
Organic Nitrogen
More than 90 percent of soil nitrogen is found in soil organic matter but often binds with soil compounds resulting in low availability to plants. Organic nitrogen becomes available when soil organic matter is decomposed by soil organisms.
Ammonium (NH+4)
Plants use ammonium (positively charged cation) that is attracted to negatively charged sites on soil particles. This form of nitrogen is available to plants, and the electrostatic attraction protects it from leaching. Ammonium converts to nitrate quickly in warm, moist soils, so soil ammonium concentrations are typically low.
Nitrate (NO3)
Negatively charged nitrate ions (anions) in soil do not react with predominantly negatively charged soil particles – keeping this form of nitrogen in soil solution and moving with water. As a result, nitrate may leach out of the root zone with excess rainfall or accumulate at the soil surface in dry conditions. Nitrate nitrogen is the form taken up by plants in the largest quantity.
Leaching -
The nitrate form of nitrogen (NO3) is very soluble and leaches easily when excess water moves through the soil. Leaching can be a major loss in coarse-textured soils, but less so in finer-textured soils. Leaching losses can be minimized by split applications to avoid high soil nitrogen during wet weather.
Nitrification
Biological oxidation of ammonium to nitrite and nitrate, or a biologically induced increase in the oxidation state of nitrogen.
Mineralization
The process of converting organic nutrients into inorganic forms that plants can use.
Immobilization
A biological process where soil microbes convert inorganic nutrients into organic compounds, making them unavailable for plants to use.