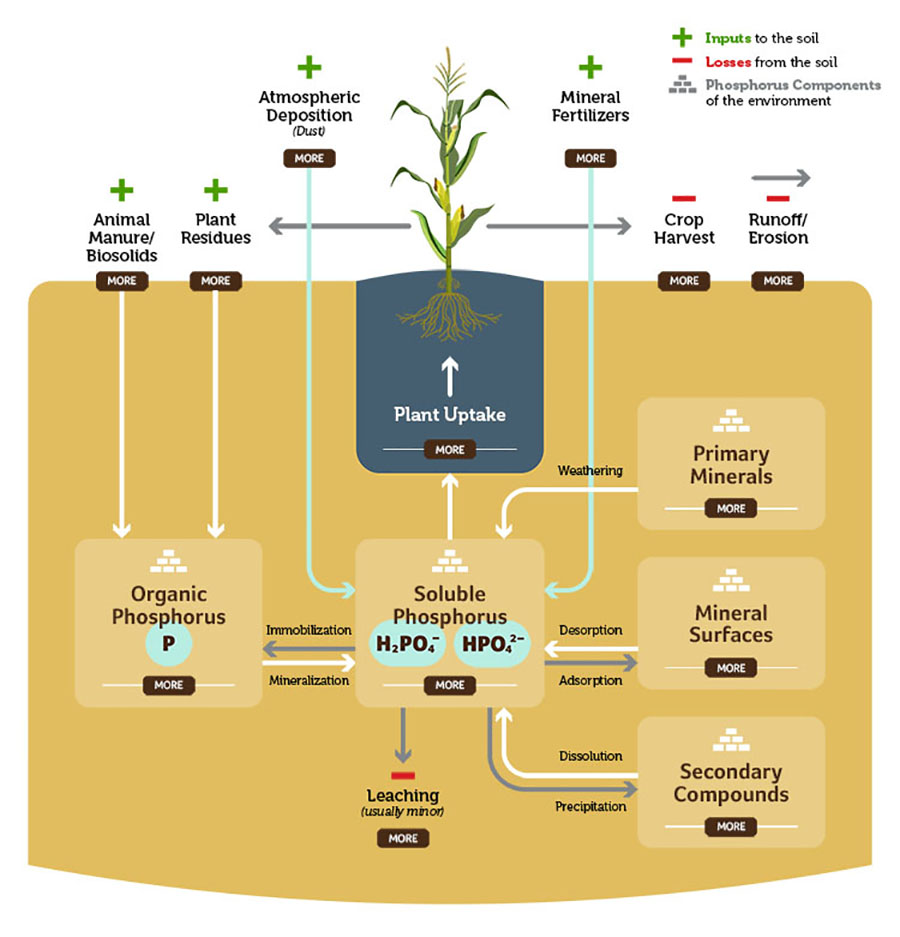

Plant Uptake
In the soil, phosphorus is in two soluble forms, H2PO4- and HPO42-. The plant’s roots absorb the phosphorus as phosphate from the soil.
Mineral Fertilizers +
Many times, depending on the need of the soil, producers will add phosphorus to their fertilizer profile. As the actual element reaches the soil and it goes through the oxidization process it is then converted into phosphate.
Animal Manure/Biosolids +
Animal manure is an important source of phosphorus – the quantity of phosphorus in the manure depends on the animal species, their age and diet, and bedding material. Once manure and biosolids are in the soil, they are considered organic phosphorus.
Plant Residues +
The crops that aren't harvested leave behind residue better known as organic phosphorus. Phosphorus in plant residues becomes available as the residues are decomposed by soil organisms.
Crop Harvest –
Since phosphorus is absorbed by the crops as phosphate, this is the amount of phosphorus that is removed from the field by harvesting the crop. Use our nutrient removal calculator to find out much phosphorus, and other nutrients, you should be replacing.
Runoff/Erosion –
Runoff is water that is not absorbed into the soil. Erosion is the wearing away of the soil through rain, irrigation, wind and ice. Because much of soil phosphorus is associated with the solid materials in soil, these actions result in phosphorus loss from the soil. Runoff and erosion can be minimized by maintaining soil cover with crop residue and/or cover crops.
Organic Phosphorus
Organic phosphorus becomes available when soil organic matter, like animal manure, biosolids and plant residues, is decomposed by soil organisms.
Soluble Phosphorus
Soluble phosphorus is the portion of soil phosphorus immediately available to plants. As plants take up soluble phosphorus, it is replenished by phosphorus from organic and mineral sources and fertilizers. Rate of this replenishment determines soil ability to meet plant needs.
Primary Minerals
The primary mineral within the soil contributing to the phosphorus cycle is a group of phosphate minerals, usual pale green to purple in color. Calcium phosphates are common in soils of slightly acid to alkaline pH. Iron and aluminum phosphates dominate in very acid soils.
Mineral Surfaces
Minerals found within the soil can affect phosphorus chemistry. Minerals within the soil can be clays, silicates, carbonates and iron and aluminum oxides and hydroxides.
Leaching
Leaching is the removal of dissolved phosphorus from soil by vertical water movement.
Secondary Compounds
Secondary compounds include calcium monophosphate, iron phosphide, manganese peroxidase and aluminium phosphide. These compounds help enhance the natural movement of phosphorus through the soil.