Fertilizer Compatibility – What You Should Know

When blending fertilizer materials there are always considerations to be made about their compatibility. There are three areas of specific consideration – chemical compatibility, critical relative humidity, and physical compatibility.
Chemical Compatibility
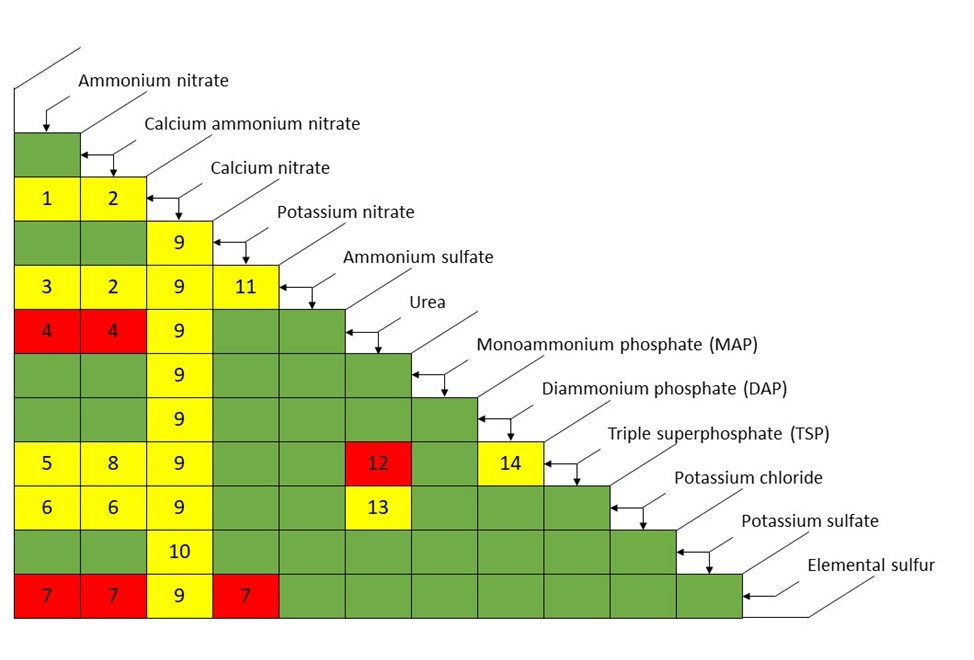
The first consideration when blending dry fertilizer materials is chemical compatibility. How will these fertilizer materials interact? Will they dissolve into a slurry? Are they safe to mix? These are all important questions. Figure 1 provides you a general guideline for blending common fertilizer materials.
The way to interpret Figure 1 is that intersections of two fertilizer materials that produce green cells are compatible and can be mixed without too much concern. Intersections of two fertilizer materials that produce yellow cells may be mixed if consideration is given to the comments that accompany the risks of mixing those materials. Intersections of two fertilizer materials that produce red cells should not be mixed.
Critical Relative Humidity
Fertilizer materials are salts and salts will absorb moisture (there are some exceptions – elemental sulfur, for instance is not a salt). This property is known as hygroscopicity. The more hygroscopic a fertilizer material, the more moisture it will absorb and the lower the critical relative humidity will be (Table 1). The critical relative humidity level is the humidity above which a fertilizer material or blend will absorb moisture from the air. Critical relative humidity is impacted by air temperature, and as air temperature increases the critical relative humidity decreases
Blending fertilizer materials will usually result in a lower critical relative humidity than the critical relative humidity of the individual fertilizers used to make the blend. For example, blending urea (73 percent) and monoammonium phosphate (92 percent) results in a blend that has a critical relative humidity of 65 percent. This can result in caked product or even slurry formation in some conditions.
Consider the blending conditions (and perhaps storage conditions) and the critical relative humidity when making dry fertilizer blends.
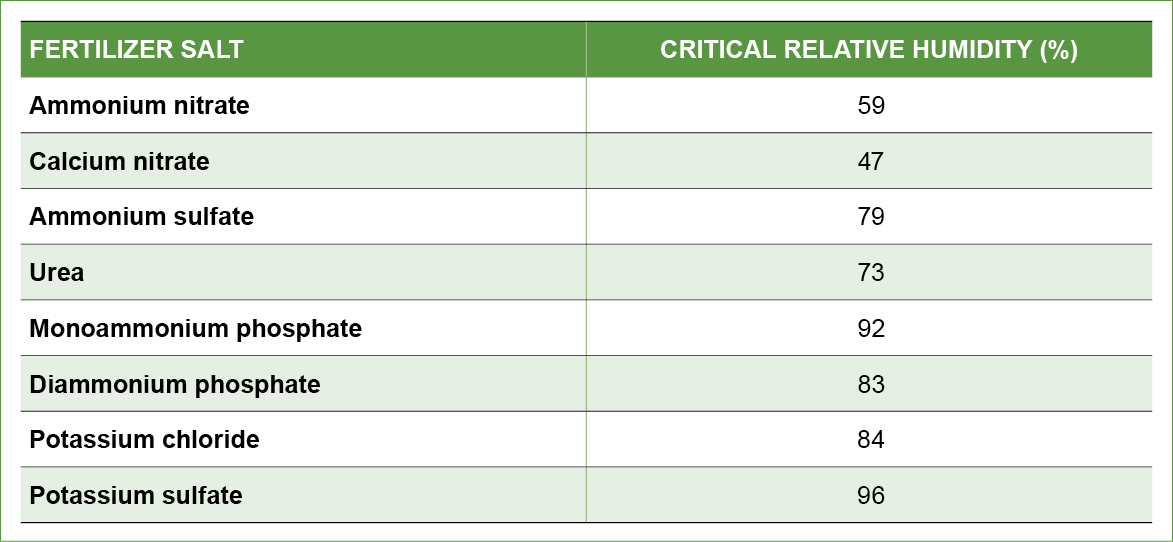
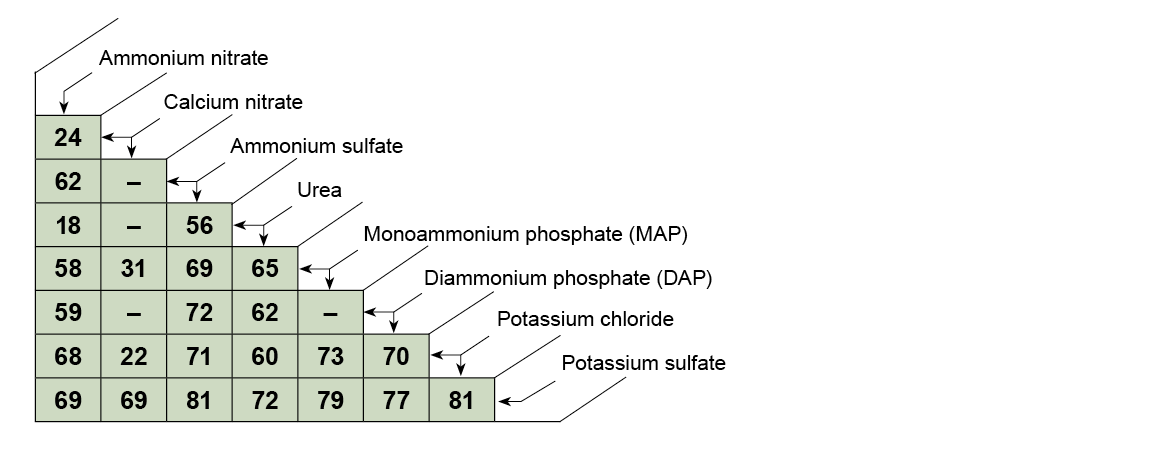
Table 1. The critical relative humidity of common dry fertilizers at 30°C
Physical Compatibility
Beyond chemical considerations and the hygroscopic nature of the blend, considerations must be made for the physical blending compatibility of dry fertilizer products. Dry products of different sizes (and even shapes and density) will determine their compatibility. Ideally, products will have the same shape and similar size. Materials of different sizes will segregate quite easily as smaller materials will settle to the bottom of the storage/applicator bin. Materials more than 10 percent different in size are likely to segregate in transport. Materials with large density differences are also more likely to segregate. This can be particularly problematic for micronutrient fertilizers, most of which have high densities and are added in small quantities.
All manufacturers of fertilizer materials should provide you with a particle size (screen guide number – SGN) and particle size distribution (uniformity index – UI). These two values are independent of one another.; Figure 3 provides a visual of three materials that have the same SGN but different UIs. Materials that have more particles close to the same average size have a higher UI. A high UI is illustrated by the first product in Figure 3 (SGN – 230, UI – 52). Notice that this material has a tight distribution of particle size. The last product in Figure 3 (SGN – 230, UI – 19) has much broader distribution of particle size.
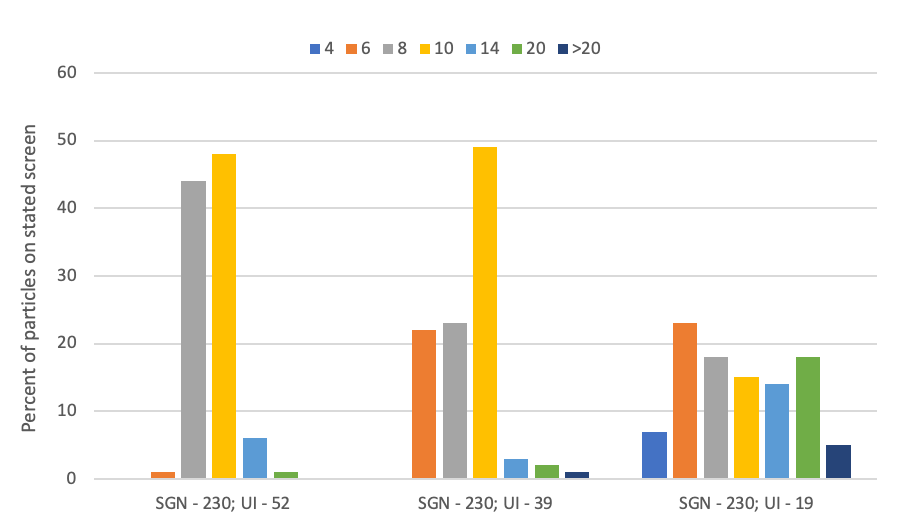
We can use the SGN and UI values to determine if fertilizer materials can be mixed without dramatic segregation. The process is relatively simple.
Let’s assume we are blending three fertilizer products (Table 2).
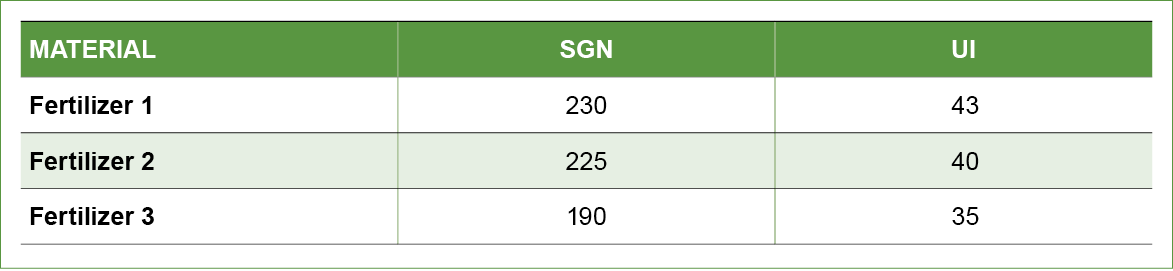
Table 2. Three dry fertilizer materials and their corresponding SGN and UI values.
We compute the average SGN and UI for the three products being considered for the blend (Table 3). Finally, we calculate 10 percent of the average to determine the acceptable ranges of SGN and UI that can be blended without dramatic segregation (Table 3).
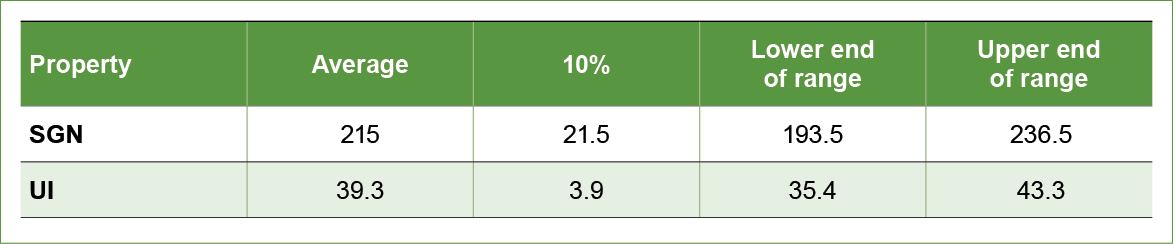
Table 3. The average SGN and UI of the three fertilizer materials being considered for a blend, as well as the lower and upper end of the acceptable range that can be blended.
Evaluation of the acceptable range of SGN and UI that will result in a non-segregating blend reveals that fertilizer 3 is not a material that should be blended with the other two products. Blending these three materials will result in fertilizer material 3 segregating from the blend resulting in poor product distribution and performance when field applied.
Make certain that fertilizer materials are physically compatible before blending.
For more information please check out these other informational sources:







