Liquid Fertilizer Mixing Compatibility – What You Should Know

Blending liquid fertilizer materials can be an effective way to put multiple nutrients into a single fertilizer blend. The question is – what liquid materials are compatible? Additionally, can one test liquid blends to determine whether potential incompatibilities exist?
Chemical Compatibility
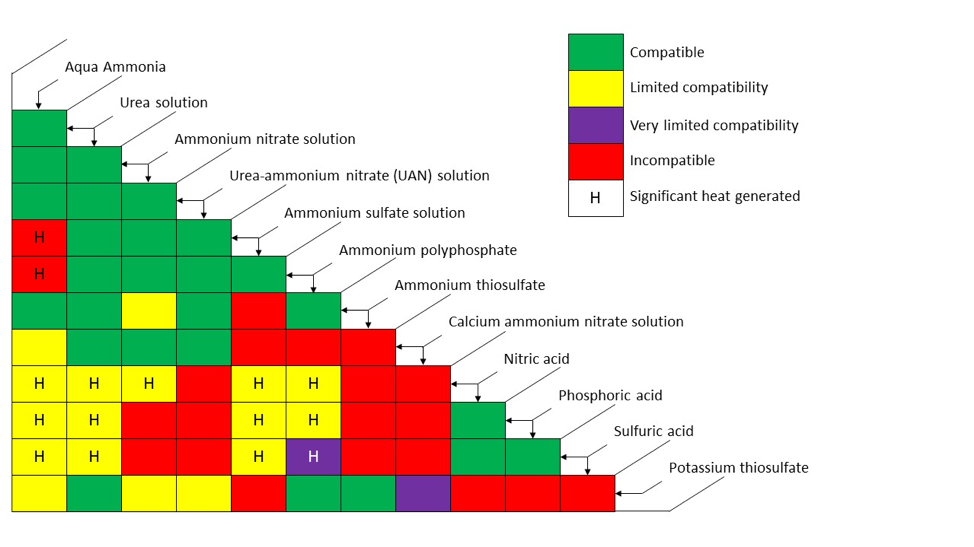
Liquid fertilizer blend compatibility is typically dictated by how much salt will stay in solution. When salt concentration gets too high, salt no longer stays in solution and solids (precipitates) will form. Figure 1 provides some general guidelines for certain liquid fertilizer blends. For example, one can see that most pure nitrogen solutions are compatible with other pure nitrogen solutions, but solutions containing phosphates, sulfur, and/or calcium have more potential for undesirable reactions. Phosphorus and calcium solutions are particularly problematic as are calcium and sulfur combinations as those mixtures form insoluble or only slowly soluble minerals.
Green cells that represent the intersection of two liquid fertilizers indicate that blends of those materials are considered compatible. Intersections of fertilizer materials that produce a yellow cell indicate limited compatibility. Limited compatibility does not necessarily mean that stable blends cannot be manufactured, but it does mean that special attention should be paid to the solubility of the mixture. So, for example, the blend may be stable, but only a specific fraction of the two materials can be successfully mixed.
Purple cells represent mixtures that have very limited compatibility. Again, this does not mean that a stable mixture of the two cannot be achieved, but additional considerations must be made (such as managing considerable heat being produced, which is potentially hazardous). Red cells represent incompatible blends due to salt-out issues, or the combination is hazardous. Cells that have an ‘H’ indicates that the blend produces considerable heat during the blending process. This can generally be managed by controlling the speed of introduction of the liquid materials (slower introduction allows for heat dissipation avoiding hazardous levels being produced).
Testing Compatibility
If uncertainty exists whether two (or more) products are compatible, bench/lab tests can/should be conducted. Bench testing of liquid compatibility does not require complicated equipment. All one really needs is the ability to measure quantities with some accuracy so that appropriate ratios of materials can be mixed to represent the larger volume to be created. If the mixture produced is stable after the initial blending, the material should be observed for several days (even weeks or months – depending upon the planned storage duration) to see if solids form in the solution. Additionally, if storage is expected to occur in colder conditions, the blended material should be exposed to the lowest expected temperature to determine if salt-out will occur during storage.
For more information:







