The Chemistry Of Soil pH

Introduction
This is part two of a three-part series taking a deep dive into soil pH.
Soil pH is a critical factor in plant and soil health. Soil pH strongly influences soil productivity; it affects a large range of soil functions such as nutrient uptake, soil microbial activity, pesticide efficacy, the ability of legumes to fix nitrogen, and disease susceptibility. Understanding why this happens is an important tool in managing the issue. And knowing chemistry of the soil pH helps to understand some of the soil reactions.
“Soil pH is considered to be the ‘master variable’ of soil chemistry due to its profound impact on countless chemical reactions involving essential plant nutrients, phytotoxic elements, and pollutants. Either directly or indirectly, pH influences the solubility of these elements determining their biological availability and mobility.” – A Critical Review on Soil Chemical Processes That Control How Soil pH Affects Phosphorus Availability to Plants.1
What is Soil pH?
Soil pH is a measure of the soil’s acidity or alkalinity – a measure of the hydrogen ion concentration. As hydrogen ion concentration increases, pH decreases. Soil pH is expressed on a scale of 1 to 14, with 1 being most acidic and 14 being most alkaline. Because pH is a negative logarithm scale, low numbers indicate greater hydrogen ion concentration. Each unit difference in pH represents a ten-fold change in hydrogen ion concentration, so pH 5 is ten times more acidic than pH 6.
The illustration below shows pH values for some common substances and different types of soil. Pure water has a pH of 7; at pH 7, hydrogen ions and hydroxide ions (OH–) are equal so pH is called neutral – neither acidic nor alkaline.

What influences soil pH?
When soluble organic and inorganic acids collect at a faster rate than they can be neutralized, it causes the acidification of the soil.3 When acidic cations (positively charged ions) like hydrogen and aluminum are abundant in the soil it becomes acidic, or it will be alkaline when basic cations like calcium, magnesium, potassium, and sodium are dominant.4
What are cations and what is CEC?
Purdue University’s Agronomy Guide provides a good explanation of cations and CEC:
Elements having an electrical charge are called ions. Positively charged elements are cations; negatively charge elements are anions. Most soil clay and organic matter particles have a net negative charge, which will attract and hold positively charged elements, or cations, much like the opposite poles of a magnet attract each other. Negatively charged particles will repel other negatively charged elements, or anions, as like poles of a magnet repel each other.5 These cations adsorbed on the soil solids can be freely exchanged with the soil solution. The total capacity of the soil to hold and exchange cations is called cation exchange capacity, or CEC. Cation exchange capacity is a basic property of the soil and is determined primarily by clay and organic matter content and chemistry. Cation exchange capacity is a large component of the soil’s pH buffering capacity, or resistance to pH change. Cations held on this complex help regulate soil pH.
A number of soil processes can produce acidity:
- Excessive rainfall and leaching
- Acidic parent material such as granite
- The decaying of organic matter
- Harvest of high-yielding crops
- Nitrification of ammonium fertilizers
Soil-acidification rate may be very slow in arid regions – occurring over centuries or millennia, but higher rainfall regions of more intense mineral weathering may acidify in a few years or decades. Sandy soils usually acidify more rapidly because of their low clay and organic matter content. The source of the parent material (granite vs shale or limestone) also has an impact on how acidic the soil will become. Limestone parent materials are more alkaline while granite parent materials are generally more acid. Like rainfall and mineral weathering, the effects of decaying organic matter tend to be small and accumulate over many years.
Harvesting and Soil Acidity
Harvesting crops can significantly affect soil acidity because crops absorb the basic elements, as cations, for their nutrition (calcium, magnesium, potassium). When these crops are harvested and the yield is removed from the field, some of the basic material is removed in the harvest, which can lead to increased soil acidity. Increasing crop yields will cause greater amounts of basic material to be removed. Grain contains fewer basic materials than leaves or stems and therefore soil acidity develops faster under continuous forage production than when only the grain is harvested. High yielding forages, like Bermuda grass or alfalfa, can speed up soil acidification more than other crops. Table 1 shows estimated amounts of basic cations removed in different portions of a wheat crop in terms of the lime equivalent to those cations removed.

Pools of soil acidity

As explained in the Soil Nutrient Management Guide from the University of Hawai’i there are three general pools, or sources, of acidity: active, exchangeable, or residual.
- Active acidity is the hydrogen ion concentration in the soil solution. The active pool of hydrogen ions is in equilibrium with the exchangeable hydrogen ions that are held on the soil’s cation exchange complex. This pool most readily affects plant growth. Active acidity is what is measured with a pH meter.
- Exchangeable acidity refers to the amount of acid cations, aluminum and hydrogen, adsorbed on the cation exchange sites. High soil CEC buffers the soil against pH changes. Acid soils with high CEC will require more lime to neutralize acidity.
- Residual acidity consists of aluminum and hydrogen bound in soil minerals. Residual acidity is least available of these three pools.6
Nitrogen and soil pH
The use of ammonium-nitrogen fertilizers, including manures and organic forms, is often a primary cause of soil acidification in agricultural soils. Acidity is produced when ammonium-containing materials are converted to nitrate in the soil, a process called nitrification.4 Nitrification is a normal microbial process in aerated soils. As ammonium (NH4) is converted to nitrate (NO3), the hydrogen ions from the ammonium molecule are released into the soil solution.
Nitrogen management practices that improve nitrogen-use efficiency can reduce acidification from nitrogen fertilizers. Nitrate leaching contributes to acidification because nitrate, an anion, when leached is accompanied by a cation, usually a basic cation. Plant nutrient uptake can also contribute to acidification as plants secrete a variety of organic acids as a normal part of their nutrient uptake and other physiological processes. Nitrogen fertilization should be accompanied by regular pH monitoring through soil testing and applying lime as needed.
To prevent this from happening:
- Use the recommended rate of nitrogen for your crop or pasture.
- Apply a little nitrogen often so that it is all taken up by the plants.
- Use best management practices to prevent losses.
- Use the least acidifying nitrogen fertiliser you can afford.
- Apply lime.
Nitrogen forms vary in their acid equivalent. Nitrate-nitrogen forms are less acidifying and may sometimes be used in place of ammonium-nitrogen forms.

Balancing soil pH
Knowing soil pH helps identify the chemical reactions that are likely to occur. Soil pH has significant effects on crop production through its effects on nutrient availability, crop productivity, crop protection chemical activity, and potential toxic elements.
Aluminum and manganese toxicities are important concerns in acidic soils. These elements are usually abundant in soils but exist primarily in insoluble minerals and soil clays. Their solubility increases sharply as pH decreases below about 5.5. High aluminum concentrations have a variety of toxic effects on plants, including inhibited root growth and development. In high pH soils, the most important effects are decreased availability of phosphorus and micronutrients. Soil pH above about 8.2-8.3 can indicate high exchangeable sodium, which will cause unfavorable physical changes in the soil such as clay dispersion, crusting, and poor permeability and water infiltration.4
Hidden Symptoms and Threshold Effects
Symptoms of soil acidity may not be apparent in crops. There may only be minor effects on nutrient availability. Yield decreases may not be observed until pH drops below some critical threshold for a specific crop. Once that threshold is passed, however, especially once aluminum reaches toxic levels, effects on crops can increase sharply up to complete loss of productivity. And because pH is usually variable across the landscape, effects may be seen in some parts of a field and not in others.7
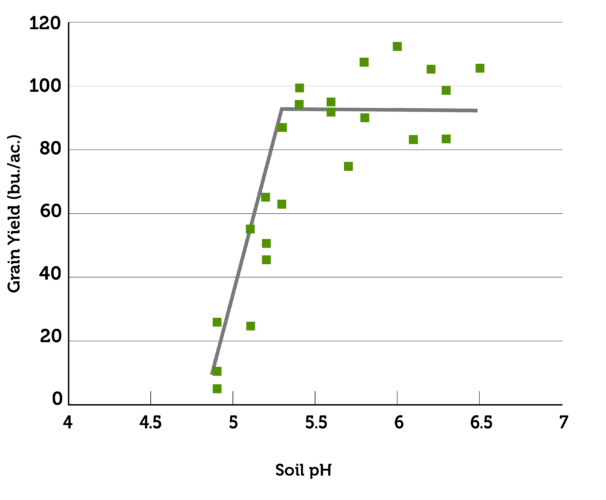
Source: Oregon State University; Figure by John Hart. Data from Kauffman, 1977.
Soil pH is an important basic property of soils that affects many aspects of plant growth and soil health. Managing soil pH must be an ongoing component of a crop-production system. Part three of this series will look at liming and other management tools for soil pH issues.
Sources:
- Penn, C.J.; Camberato, J.J. A Critical Review on Soil Chemical Processes that Control How Soil pH Affects Phosphorus Availability to Plants. Agriculture 2019, 9, 120. https://doi.org/10.3390/agriculture9060120
- A Critical Review on Soil Chemical Processes that Control How Soil pH Affects Phosphorus Availability to Plants
- Living on The Land: Managing Soil pH
- Earth and Planetary Sciences: Soil Acidification
- Oklahoma State University Extension: Organic Matter Decay
- Purdue University Extension: Buffer Capacity and Percent Base Saturation
- University of Hawaii: Pools of Soil Acidity
1 A Critical Review on Soil Chemical Processes that Control How Soil pH Affects Phosphorus Availability to Plants
2 Living on The Land: Managing Soil pH
3 Earth and Planetary Sciences: Soil Acidification
4 Oklahoma State University Extension: Organic Matter Decay
5 Purdue University Extension: Buffer Capacity and Percent Base Saturation







